 B., Thermodynamic Properties of Individual Substances, Fouth Edition, Hemisphere Pub. For one bond, the bond dipole moment is determined by the difference in electronegativity between the two atoms. Landolt-Bornstein: Group II: Volume 6 Molecular Constants from Microwave, Molecular Beam, and Electron Spin Resonance Spectroscopy Springer-Verlag. A dipole moment measures a separation of charge. Let the charges on the plates be +qand qand their areas are A. Figure 1: Scheme of a charged capacitor without (a) and with (b) dielectric. 
Consider two plane, parallel plates depicted in Fig. Please address comments about this page to KH and AM Hellwege (eds.). The dipole moment of a molecule may be obtained from measurements of the relative permittivity of a bulk sample. NIST does not necessarily endorse the views expressed, or concur with the facts presented on these sites.įurther, NIST does not endorse any commercial products that may be mentioned on these sites. There may be other web sites that are more appropriate for your purpose. No inferences should be drawn on account of other sites being referenced, or not, from this page. We have provided these links to other web sites because they may have information that would be of interest to you. Such is the case for CO 2, a linear molecule (part (a) in Figure 2.2.8). If the individual bond dipole moments cancel one another, there is no net dipole moment. Even though the two dipole moments are equal in magnitude (since they are both oxygen hydrogen bonds), they are not arranged symetrically about the central atom and thus will not cancel out.You are here: Experimental > One molecule all propertiesĮxperimental data for BrF (Bromine monofluoride)īy selecting the following links, you may be leaving NIST webspace. The dipole moment of a molecule is therefore the vector sum of the dipole moments of the individual bonds in the molecule. Carbon dioxide will not be polar because both dipole moments are equal in magnitude (since they are both carbon oxygen bonds) and arranges symetrically about the central atom in a linear geometry. Electric dipole moments of fundamental particles, such as the electron, are signatures of time-reversal symmetry violation, equivalent to violation of combined charge and parity (CP) symmetry (). Water is a bent molecule with two polar bonds. Carbon dioxide is a linear molecule with two polar bonds. Where is the distance between the two charges. Let's look at two examples: carbon dioxide and water. (i) The polarity of a covalent bond can be measured in terms of dipole moment which is defined as q×2d. In the following geometries, the atoms are arranged symetrically about the central atom: linear, triangular planar, tetrahedral, square planar, triangular bipyrimidal, and octahedral. They are symetrically arranged about the central atom. the bonds are made with the same element) ANDĢ. Any resulting dipole indicates a polar molecule.ġ. Step 2: Using the molecular geometry, determine if any of the dipole moments will cancel. You have found all of the polar bonds in each of these three molecules. A molecular vibration is a periodic motion of the atoms of a molecule relative to each other, such that the center of mass of the molecule remains unchanged. Lewis structures for H 2S, BF 3, and CCl 2H 2Īre shown below. experience a moment E EE, which by definition refers to the. This occurs because of a difference in electronegativity of the two atoms that share the electrons. Electric dipole moment, the measure of the electrical polarity of a system of charges Magnetic dipole moment, the measure of the magnetic polarity of a system. Free-Point molecular dipole interactions Suppose a set of polarizable molecules are. 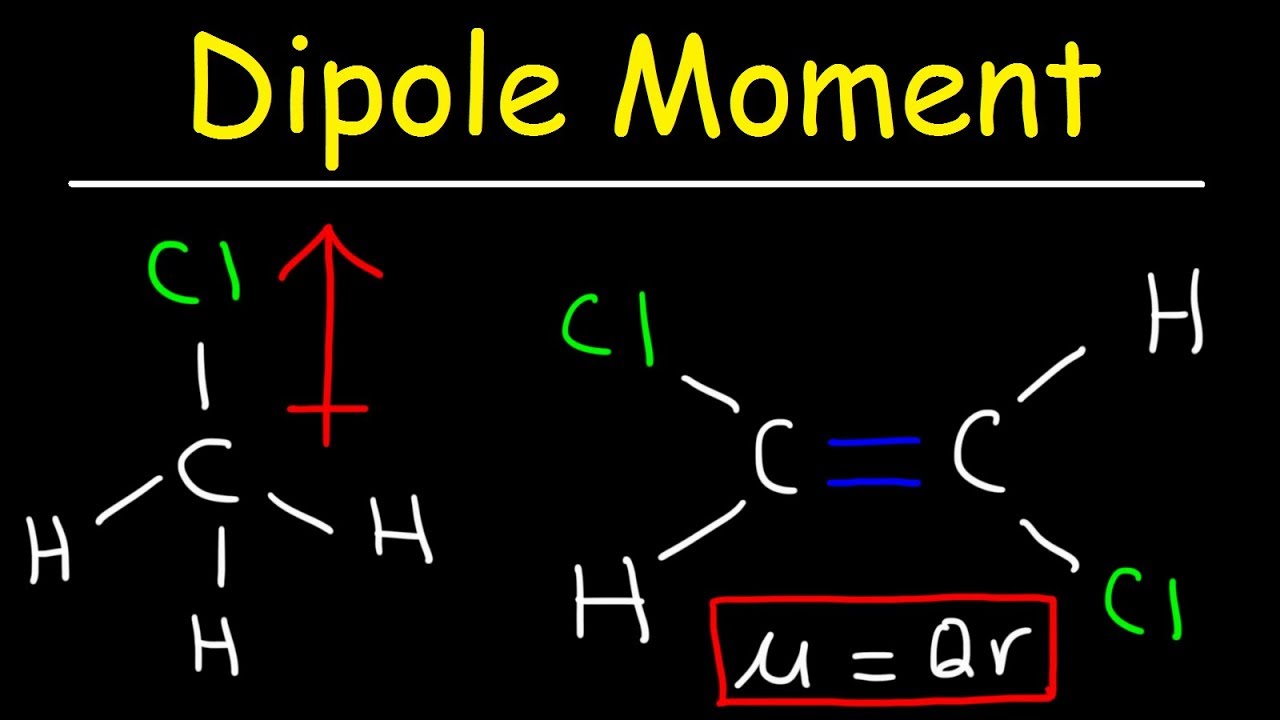
Remember that a polar bond is one in which the electrons are unevenly distributed. Step 1: Indicate polar bonds in molecule or ion. About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy & Safety How YouTube works Test new features NFL Sunday Ticket Press Copyright. As you work through these steps you will see that molecules with polar bonds are not necessarily polar molecules. 
There is a series of steps you can take to determine if a molecule is polar or not. 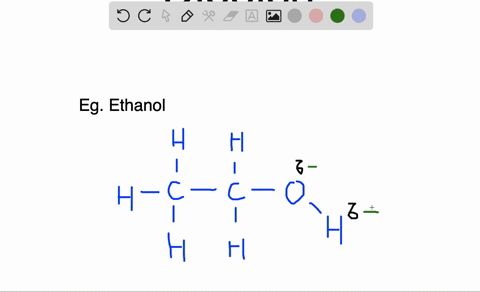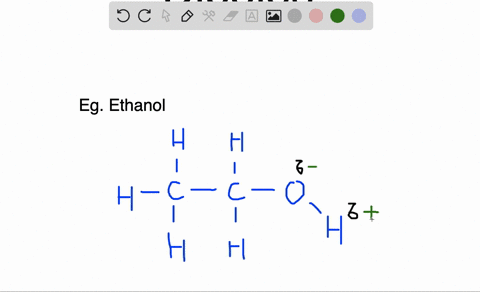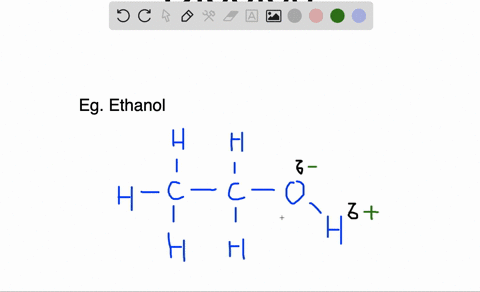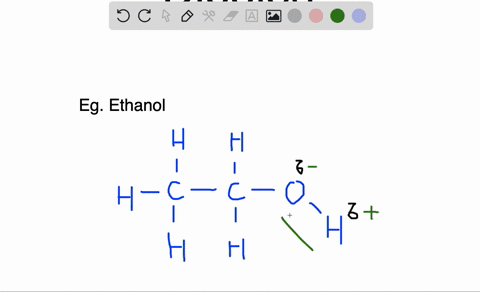
Likewise molecules in which there is an accumulation of electron density at one end of the molecule, giving that end a partial negative charge and the other a partial positive charge, are called polar molecules. The electron density of a polar bond accumulates towards one end of the bond, causing that end to carry a slight negative charge and the other end a slight positive charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
